 
Element/Moleculeīy knowing the molar mass of a substance, having any mass of that substance allows one to know how many moles of it they have. A list of some molar masses can be seen below. The molar mass of any element or molecule is given by the sum of the atomic weights multiplied by 1 g/mol. In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol. Hydrogen (H) Atomic Data for Hydrogen (H) Atomic Number 1 Atomic Weight 1.00794 Reference E95 : Isotope : Mass : Abundance : Spin : Mag Moment : 1 H: 1.007825: 99.985: 1/2 +2.79284: 2 H: 2.0140: 0.015: 1 +0.85743: Notes: The wavelengths and energy levels given below are for the isotope 1 H. This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. This article was written for you by Samantha, one of the tutors with SchoolTutoring Academy.Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance. Looking to get ready for the ACT? We can help with ACT Prep 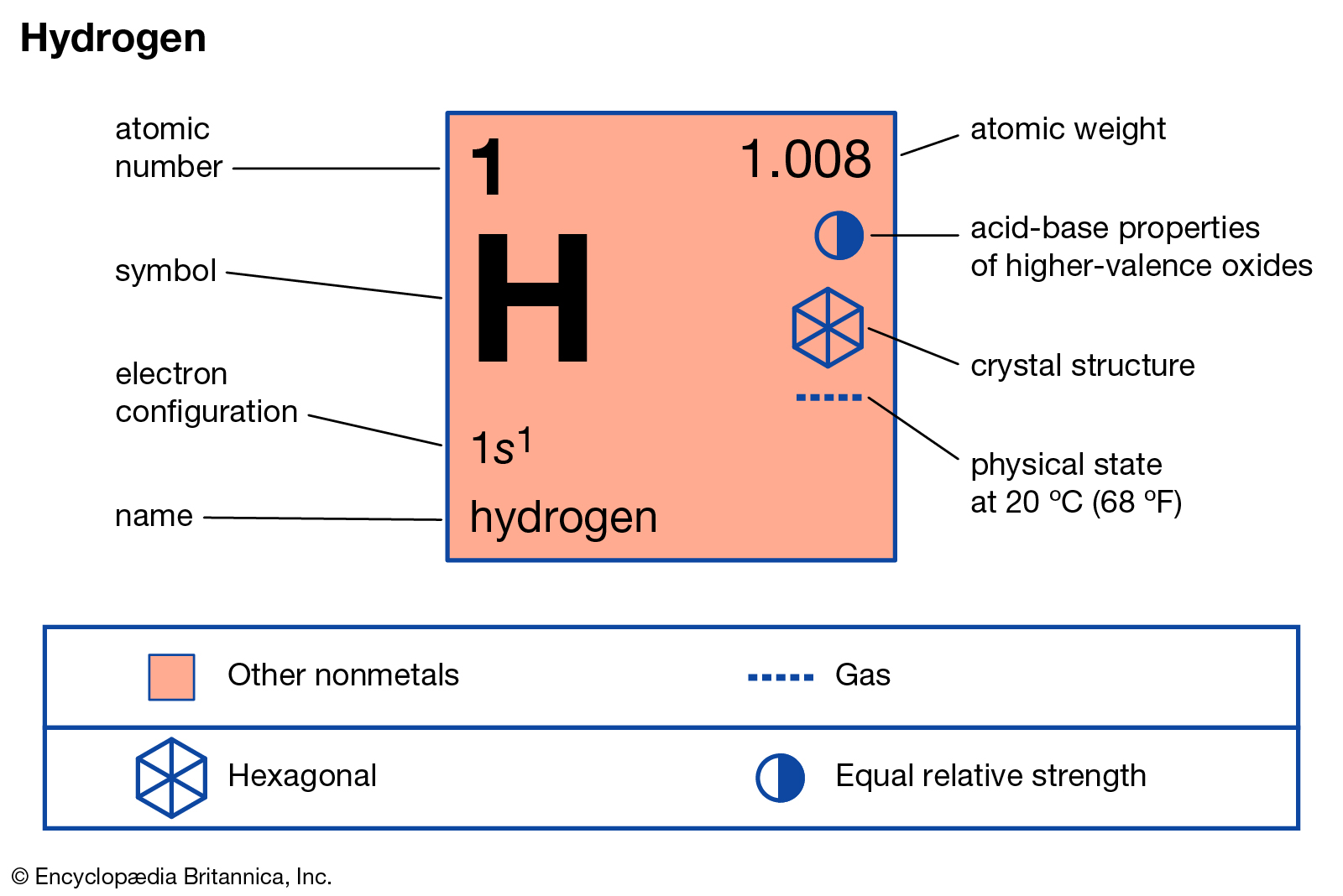
Therefore, you have 136.2652 g of calcium. Since the atomic mass of calcium is 40.078 amu, the molar mass of calcium is 40.078 g/mole.Ģ) Multiply the number of moles of calcium you have by the molar mass. Therefore, you have 0.4996 mole of nitrogen.Įxample: If you had 3.4 moles of calcium, how many grams of calcium do you have?ġ) Figure out what the molar mass of calcium is. The nucleus of tritium (t, sometimes called a triton) contains one proton and two neutrons, whereas the nucleus of the common isotope hydrogen-1 ( protium) contains one. Since the atomic mass of nitrogen is 14.01 amu, the molar mass of nitrogen is 14.01 g/mole.Ģ) Divide the amount of nitrogen you have by the molar mass. The relative atomic mass minus the atomic number tells you there are no neutrons in the nucleus of an average atom. Tritium (from Ancient Greek (trtos) 'third') or hydrogen-3 (symbol T or 3H) is a rare and radioactive isotope of hydrogen with a half-life of about 12 years. The molar mass of water would then be 18.016 g/mole.Įxample: If you had 7 g of nitrogen, how many moles of nitrogen do you have?ġ) Figure out what the molar mass of nitrogen is. 
To get the molar mass of water, you would add up the molar masses of 2 hydrogens (2 x 1.01 g) and 1 oxygen (1 x 16 g). For example, a water molecule is made up of 2 hydrogen atoms and 1 oxygen atom. It is approximately equal to the number of protons and neutrons, with a little extra added by the. To get the molar mass for a compound, you add up all the molar masses of the atoms in the compound. The total weight of an atom is called the atomic weight. Thus, the molar mass of hydrogen is 1.01 g/mole. If hydrogen is the lightest of all substances, then why not give it a. This means that 1 mole of hydrogen is 1.01 g. If you check a periodic table, youll see that Hydrogen actually has a mass of 1.00794. Thus, 1 mole of any element has a mass in grams that is numerically equivalent to its atomic mass.įor example, the atomic mass of hydrogen is 1.01 amu. 12 grams is equal to 1 mole of carbon-12, which has 6.02×10 23 atoms. If we look at 1 atom of carbon-1, it has a mass of 12 amu. Molar mass is used to convert moles to grams. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
